16 January
WORLD SNOW DAY

„Save the date” is a series of articles that have been written to celebrate various unusual holidays. The authors of the presented materials are students, doctoral students and employees of the Faculty of Science and Technology of the University of Silesia.
16 January is World Snow Day.
Anna Maroń, PhD, Joanna Palion-Gazda, PhD and Prof. Barbara Machura | Institute of Chemistry
For centuries, people have been interested in the mystery hidden behind snowflakes falling gently outside their windows on cold winter days. Single ice crystals known as snowflakes are formed high above our heads from water vapour, creating unique, often very spectacular, but always hexagonal forms, which results from the structure of hydrogen bonds forming in the ice crystal structure.

The crystal structure of ice.
However, it is not only the sheer beauty of this phenomenon that attracts people. Snowflakes are the solid form of water necessary for life on our planet. Their formation and presence is crucial for many meteorological and environmental phenomena. They play an important role in the process of electrification of clouds, electrical discharges, and they are also important in chemical processes occurring in the higher layers of the atmosphere, for which the contact surface with the ice layer is crucial. The study of the formation and growth dynamics of snowflake crystals is a fundamental problem in science.
The first scientific view of the symmetry of geometric forms of snowflakes was given by Johannes Kepler in his work On the Six-Cornered Snowflake: A New Year’s Gift (1611). The philosopher finds many analogies between the hexagonal shapes of snowflakes and honeycombs and considers his problem on the basis of the theory of compact, hexagonal arrangement of spheres in space. The New Year’s Gift is considered to be the first treatise ever to ponder the problems of crystallisation, and perhaps even the first scientific paper on crystallography. Twenty-six years later, in his work Les Metéores, Rene Descartes gave a description of the morphology of snowflakes, and Robert Hooke in 1665 published a volume entitled Micrographia, in which he included sketches of things he viewed under the microscope, including drawings of snowflakes. Further progress in this area came with the development of photography. In 1931, Wilson Bentley, who became known as “The Snowflake Man”, published his famous book Snow Crystals, which is still being reissued today, featuring over 2000 microphotographs of snowflakes. It was these photographs that inspired physicist Ukichiro Nakaya to begin systematic laboratory research into the process of ice crystal growth. He investigated how various factors such as temperature and air humidity affect the form of snowflakes. This led to the creation of the snowflake morphology diagram.
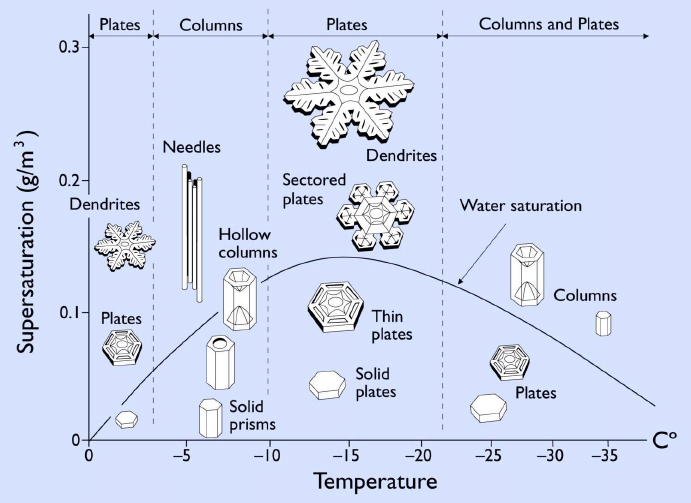
Snowflake morphology diagram (Kenneth G. Libbrecht, Caltech).
The growth process of a single snowflake begins when a water vapour droplet in a cloud encounters, for example, a dust particle in the air. This provides the starting point for the crystal nucleation process. When the temperature drops, the phase change of the water vapour into solid, i.e. ice, begins. In the initial phase, only hexagonal plate-like crystals are formed, which can then take on a variety of forms during the growth process.
Although now many aspects related to the snowflake growth process are well understood, such as the crystal structure of ice, the nature of interactions between water molecules, the phase transition diagram for water, or the equilibrium structure of the ice surface, some fundamental aspects related to the snowflake growth phenomenon, such as the snowflake morphology diagram, still have many secrets yet to be uncovered!
On these beautiful winter evenings, let’s not let ourselves take away from the childlike view of snowflakes that have fascinated us for centuries. Let us find a moment to admire their beauty, while quietly humming “Little snowflake falling from the sky”.
References:
- Johannes Kepler, Noworoczny podarek albo o sześciokątnych płatkach śniegu, Wydawnictwo Uniwersytetu Warszawskiego, Warszawa, 2006.
- Kenneth G. Libbrecht Rep. Prog. Phys. 2005, 68, 855.



Photos of differently shaped snowflakes (Wilson A. Bentley)